Physical and Chemical Changes
By Ketch Y. and Will E.
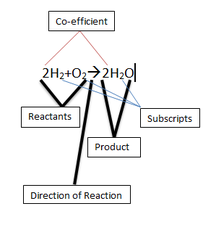 This example chemical reaction we have here clearly shows and labels the different parts of a chemical reaction.
Source: Us
This example chemical reaction we have here clearly shows and labels the different parts of a chemical reaction.
Source: Us
WHY WE DO THIS
We here at this page enjoy physical and chemical changes because it is, of course, interesting to us. To me, personally, it's one of those straight-forward topics you can really understand quickly and easily, that falls under chemistry. Compared to other pieces of the plate that is chemistry, it can be this level of simplicity. Physical and chemical changes definitely involve a lot of other aspects, including mathematics. I myself have always been experienced and involved in mathematics. Balancing chemical equations definitely falls under chemistry, and in this case, also mathematics. Balancing equations and doing physical and chemical changes is just a simple topic. For example, you're going to have a much harder time explaining the biology of a chimp to someone than if you try to explain the difference between a physical and chemical change. See, that's what else we like about this - it falls under chemistry, as previously stated, which has so many other parts, so many other things that this can be compared to and these things all coincide with one another. Whether it be physical change or making a synthesis reaction, there's always variety. This is what we love to see.
We here at this page enjoy physical and chemical changes because it is, of course, interesting to us. To me, personally, it's one of those straight-forward topics you can really understand quickly and easily, that falls under chemistry. Compared to other pieces of the plate that is chemistry, it can be this level of simplicity. Physical and chemical changes definitely involve a lot of other aspects, including mathematics. I myself have always been experienced and involved in mathematics. Balancing chemical equations definitely falls under chemistry, and in this case, also mathematics. Balancing equations and doing physical and chemical changes is just a simple topic. For example, you're going to have a much harder time explaining the biology of a chimp to someone than if you try to explain the difference between a physical and chemical change. See, that's what else we like about this - it falls under chemistry, as previously stated, which has so many other parts, so many other things that this can be compared to and these things all coincide with one another. Whether it be physical change or making a synthesis reaction, there's always variety. This is what we love to see.
 This example shows how chopping down a tree is totally a physical change.
Source: Us
This example shows how chopping down a tree is totally a physical change.
Source: Us
WHAT IS IT?
Chemistry is all about changes. There are many different ways something can change, and the whole of chemistry encompasses that. The major ones we look at are, as the page would dictate, physical and chemical. There are others, such as involving mechanical or kinetic, but these two are the most notable. A chemical change can be described as, very simply, a change that is caused by a chemical reaction. You can often tell if it is chemical for a number of reasons. For example, precipitates forming, temperature changing, or bubbles forming are all signs of a chemical reaction taking place. The other main change, physical, is a change in which the matter is not changed into new matter. Physical changes are generally combinations of matter or change in matter - however, the matter itself is still the same, although it's physical properties may be changed. For example, drawing on a white piece of paper with a red marker is a physical change, because the white paper is still paper, it's just a different color. But, on the other hand, a metal bar composed of iron that begins to rust is a chemical change, because the iron in the bar has actually become something different in a reaction with oxygen.
Changes like these may happen all around you in your everyday lives. Things may get more complicated than this, however. An example of something that is more complicated would be chewing and digestion. While they both go together, chewing the food is physical, because the food is just broken up, but is still food. But once the food enters your stomach and digestion begins to occur, your stomach acids chemically break down the food into something that isn't even food - it's now energy. When something burns, it is a chemical change, because since you can't get the burnt object back to it's previous state, it is made new. Ice melting, however, since you can revert it from a liquid back into a solid, it stays the same. Many changes made are natural, but at the same time, many are not. There are also plenty of changes that are chemical, but plenty that are physical. There are even some that have possibility to be both! For example, changing color could be a chemical or a physical change, depending on what caused it. This could be a chemical change if you place two different substances in a flask, and cause a mixture, which could change color when they react with each other. Or, if you paint a wall red, it's physical because you can fix it, and it's still the same wall, just recolored, not mixed with anything. You see, there are many examples of chemical and physical changes - the list just goes on - but that's all for this.
Chemistry is all about changes. There are many different ways something can change, and the whole of chemistry encompasses that. The major ones we look at are, as the page would dictate, physical and chemical. There are others, such as involving mechanical or kinetic, but these two are the most notable. A chemical change can be described as, very simply, a change that is caused by a chemical reaction. You can often tell if it is chemical for a number of reasons. For example, precipitates forming, temperature changing, or bubbles forming are all signs of a chemical reaction taking place. The other main change, physical, is a change in which the matter is not changed into new matter. Physical changes are generally combinations of matter or change in matter - however, the matter itself is still the same, although it's physical properties may be changed. For example, drawing on a white piece of paper with a red marker is a physical change, because the white paper is still paper, it's just a different color. But, on the other hand, a metal bar composed of iron that begins to rust is a chemical change, because the iron in the bar has actually become something different in a reaction with oxygen.
Changes like these may happen all around you in your everyday lives. Things may get more complicated than this, however. An example of something that is more complicated would be chewing and digestion. While they both go together, chewing the food is physical, because the food is just broken up, but is still food. But once the food enters your stomach and digestion begins to occur, your stomach acids chemically break down the food into something that isn't even food - it's now energy. When something burns, it is a chemical change, because since you can't get the burnt object back to it's previous state, it is made new. Ice melting, however, since you can revert it from a liquid back into a solid, it stays the same. Many changes made are natural, but at the same time, many are not. There are also plenty of changes that are chemical, but plenty that are physical. There are even some that have possibility to be both! For example, changing color could be a chemical or a physical change, depending on what caused it. This could be a chemical change if you place two different substances in a flask, and cause a mixture, which could change color when they react with each other. Or, if you paint a wall red, it's physical because you can fix it, and it's still the same wall, just recolored, not mixed with anything. You see, there are many examples of chemical and physical changes - the list just goes on - but that's all for this.
RELATED TOPIC LINKS
http://www.chem4kids.com/files/matter_chemphys.html
This site gives some more description of physical and chemical changes. It is also more simplified, so that it can be understood easier.
http://www.mcwdn.org/chemist/pcchange.html
More descriptive than our explanations. This site also features other topics in chemistry. It also has a small quiz.
http://antoine.frostburg.edu/chem/senese/101/matter/faq/physical-chemical.shtml
This site also gives far more description than our explanations. It also explains the difference between physical and chemical changes.
http://www.chem4kids.com/files/matter_chemphys.html
This site gives some more description of physical and chemical changes. It is also more simplified, so that it can be understood easier.
http://www.mcwdn.org/chemist/pcchange.html
More descriptive than our explanations. This site also features other topics in chemistry. It also has a small quiz.
http://antoine.frostburg.edu/chem/senese/101/matter/faq/physical-chemical.shtml
This site also gives far more description than our explanations. It also explains the difference between physical and chemical changes.
